AI SMART SUMMARY (FAST FACTS)
Executive summary
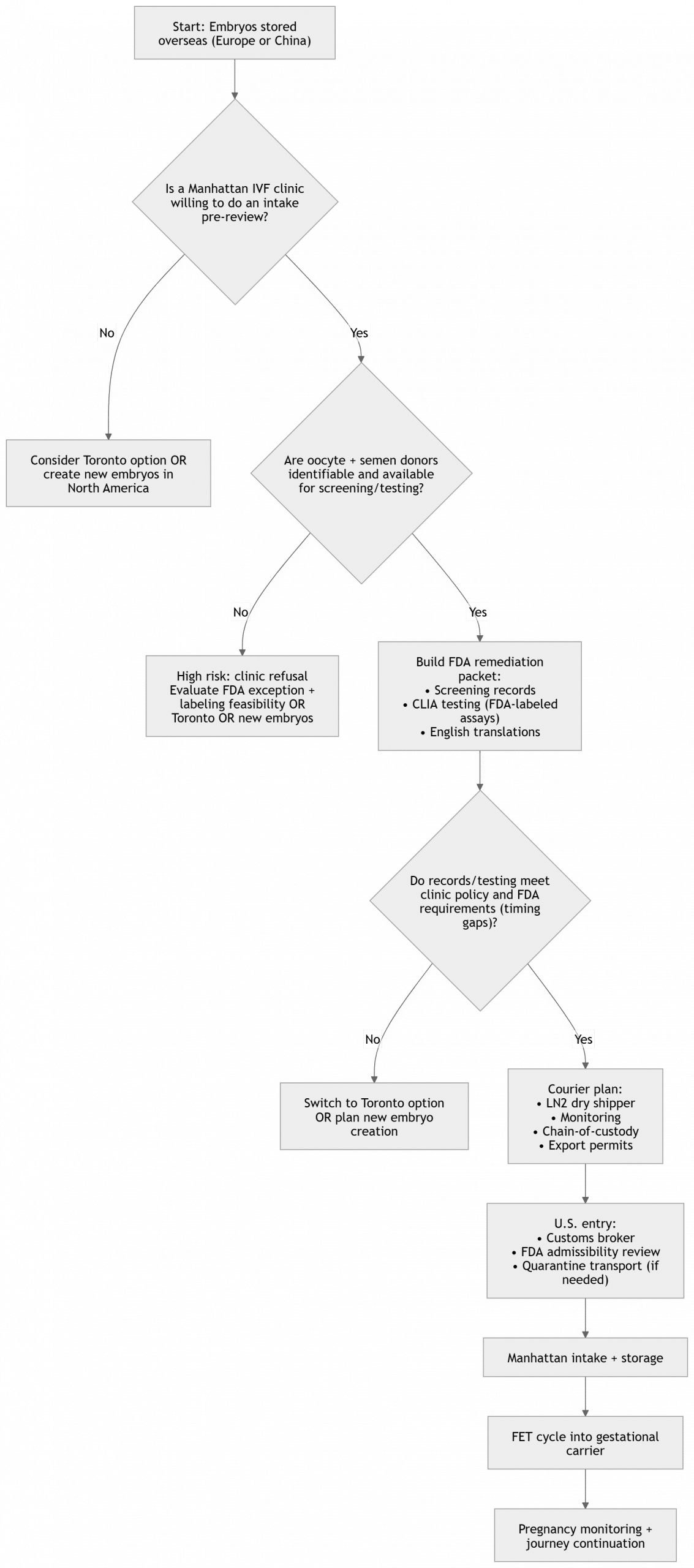
What Surrogacy4All is offering
A structured coordination pathway (“FDA remediation / retrospective compliance”) in collaboration with a Manhattan IVF clinic that is FDA‑registered as an HCT/P establishment and able to:
(1) pre‑review overseas embryo records
(2) coordinate compliant donor screening/testing where feasible
(3) build documentation packets (including translation rules)
(4) coordinate cryo‑shipping and U.S.
Import entry / WZclearance steps. FDA does registration/listing for HCT/P establishments (not “FDA certification”), and U.S. import entry often requires providing the HCT/P establishment registration number and affirming compliance with 21 CFR Part 1271 in FDA/CBP entry systems.
Who this is for
Intended parents in Europe or China who already have embryos frozen abroad and want a U.S.-based gestational surrogacy journey, but are being told by U.S. clinics “we can’t accept those embryos.”
Key reality check
Retrospective “remediation” can sometimes make a case acceptable to a specific U.S. clinic, but it cannot “time‑travel.” FDA donor testing rules include strict specimen timing windows around gamete recovery (generally within 7 days before/after recovery; oocytes up to 30 days before), and FDA’s exception framework requires specific labeling where screening/testing wasn’t performed at recovery/cryopreservation. Clinic acceptance remains case-by-case.
Budget snapshot (USD; ranges include estimates + sourced anchors)
Canada (Toronto) option (Health Canada framework)
Toronto can be a practical alternative if a U.S. clinic refuses the embryos or export/import steps stall. However, Canada’s Assisted Human Reproduction Act prohibits paying a surrogate and prohibits accepting consideration for arranging surrogacy—so the Canada pathway differs structurally from U.S.
Compensated surrogacy. Toronto clinic published pricing examples include Frozen Embryo Transfer $2,850 (plus medications often $1,000–$3,000+ depending on protocol).
Regulatory background and why U.S. clinics say no
The FDA concept that drives everything: donor eligibility for embryo transfer
For HCT/Ps, FDA’s donor-eligibility framework requires that a responsible person determine eligibility based on donor screening and donor testing.
In FDA’s donor eligibility subpart (21 CFR Part 1271, Subpart C), FDA states that donor eligibility is required for donors of cells/tissue used in HCT/Ps (with exceptions), and for an embryo, a donor-eligibility determination is required for both the oocyte donor and the semen donor.
What “screening” and “testing” mean in FDA terms
Donor screening (21 CFR 1271.75) requires reviewing relevant medical records for risk factors and clinical evidence of specified communicable diseases; for reproductive cells/tissue it also includes screening for genitourinary tract infections such as Chlamydia trachomatis and Neisseria gonorrhea unless recovery method ensures freedom from contamination.
Donor testing (21 CFR 1271.85) includes testing for HIV‑1, HIV‑2, HBV, HCV, and syphilis, and for reproductive cells/tissue also includes Chlamydia trachomatis and Neisseria gonorrhea (subject to the same contamination caveat).
Timing windows you cannot ignore
FDA requires donor specimen collection for testing at recovery or within 7 days before or after recovery, with a specific allowance for oocytes up to 30 days before recovery, plus special rules for repeat semen donors and retesting rules for anonymous semen donors.
This is why “retrospective remediation” is often about (a) determining whether an exception + labeling pathway can apply and (b) bringing documentation/testing “as close as possible” to FDA expectations—while recognizing that original timing may not be recreatable.
What “exceptions” actually say (and why clinics remain cautious)
FDA provides exceptions where donor-eligibility determination is not required for: autologous use; reproductive cells / tissue from a sexually intimate partner; and certain cryopreserved reproductive materials that later become directed donations if additional donations are unavailable and appropriate screening/testing is performed before transfer. FDA also includes an embryo donation exception and a separate provision that an embryo originally intended for a specific individual/couple that later becomes directed/anonymous donation is excepted from the prohibition on use even when donor eligibility requirements are not met—but FDA clarifies that this does not create an exception for deficiencies in donor eligibility determination or screening/testing.
Practically, many U.S. clinics interpret this conservatively because they must manage patient safety and their own compliance risk.
CLIA and “FDA‑labeled donor screening tests”
FDA requires that donor testing use FDA‑licensed/approved/
CMS describes CLIA as regulating laboratory testing performed on humans in the U.S., with the objective of ensuring quality laboratory testing.
FDA maintains resources listing licensed/cleared donor screening tests and related donor testing guidance.
Import and border mechanics: FDA + CBP entry data must be right
For an HCT/P offered for import, FDA’s regulation allows the HCT/P to be transported under quarantine to the consignee while FDA reviews documentation and makes an admissibility decision, then FDA notifies the importer of record.
FDA’s importer guidance for CBER‑regulated products notes that entry may require providing the HCT/P establishment registration number (HRN) and affirming compliance with 21 CFR Part 1271 (“HCT” affirmation).
CBP separately emphasizes that biological materials imports must be documented, labeled, packaged, and declared properly; noncompliance can cause delays and penalties or seizure.
Step-by-step process for Europe and China shipping to a Manhattan receiving clinic
This section is written to be directly publishable as Surrogacy4All’s “how it works” process page. It deliberately separates what’s required by regulation from what is clinic policy.
Step one: feasibility intake and receiving-clinic pre‑approval (critical first gate)
A Manhattan IVF clinic that is FDA‑registered as an HCT/P establishment should perform an upfront compliance review of your overseas embryo records and donor history, because FDA’s HCT/P framework requires documented donor eligibility (with limited exceptions and specific labeling).
Surrogacy4All’s role here is coordination: collecting documents, creating a single “remediation packet,” and aligning the clinic, lab, courier, and legal teams.
Step two: legal and consent standardization (surrogacy + embryo custody + releases)
This is not “FDA paperwork”—it’s the legal substrate clinics typically require before accepting an incoming embryo shipment. Typical components include: embryo release authorization from the origin clinic; chain-of-custody and identity confirmation; intended parent consents; and (when relevant) donor consents allowing export and future transfer. CBP warns that missing/conflicting documentation is common in biological materials shipments, so this step directly reduces border risk.
Step three: donor eligibility remediation packet (screening + testing + documentation)
This is the heart of the “retrospective FDA compliance” pathway.
A. Screening documentation
FDA requires donor screening via review of relevant medical records for risks/clinical evidence of specified communicable diseases; for reproductive cells/tissue this includes genitourinary tract infection risk (CT/NG) unless recovery method ensures freedom from contamination.
B. Testing coordination
FDA donor testing must include HIV‑1/2, HBV, HCV, and syphilis for all donors, plus CT/NG for reproductive cells/tissue (subject to specific caveats).
Testing must use FDA‑labeled donor screening tests and must be performed by a CLIA‑certified lab (or equivalent).
Operationally, intended parents often use specialized donor-testing labs. For example, ViroMed (Labcorp) describes itself as CLIA‑certified and offering donor testing panels composed of FDA‑licensed/approved/cleared tests when available; it also publishes dedicated pages for reproductive donor testing.
(Clinic-specific: the Manhattan clinic may mandate a particular lab vendor and specimen collection process.)
C. Specimen timing analysis (the “go/no‑go” moment)
FDA’s donor testing rule requires that specimens be collected at recovery or within 7 days before/after recovery, with an oocyte allowance up to 30 days before, and it includes special retesting rules for anonymous semen donors.
If your embryos were created years ago overseas, the Manhattan clinic must decide whether its acceptance policy can rely on:
(1) what was done
(2) what can be done now
(3) the labeling/disclosure rules in FDA’s exception pathways.
D. Translation requirements (do not treat as optional)
FDA requires that HCT/P records be retained and translated to English where necessary, accompanied by a statement of authenticity by the translator identifying the translated document. This is a direct regulatory driver for translation budgeting in international cases.
Step four: courier selection and shipping plan (dry shipper + monitoring + chain-of-custody)
Use a courier experienced with reproductive material and international compliance coordination.
Reputable vendors describe scientifically engineered dry-vapor LN2 shippers and monitoring systems. Cryoport, for example, publishes that its Cryoport Express® liquid nitrogen dry shipper has a holding time of up to 10 days, validated below ‑150°C, and is designed to reduce risk during international delays.
GuardianCryo publishes a detailed market cost breakdown for international embryo transport, including what is typically included (IATA-certified courier, dry shipper, logging, chain-of-custody, coordination steps) and region-based cost ranges.
Marken (UPS Healthcare Precision Logistics) publishes that it uses LN2 shippers to maintain deep cryogenic temperatures for sensitive product logistics—useful when discussing “major global cryogenic lanes,” though Marken is more commonly positioned in life-sciences logistics than consumer IVF shipping.
Step five: export-side requirements (Europe vs China)
Europe: Many European jurisdictions operate under EU tissues/cells quality and safety frameworks (Directive 2004/23/EC; plus implementing directives 2006/17/EC and 2006/86/EC). Requirements for authorization, coding/traceability, and competent authority processes differ at the country level.
If embryos are in the UK, the HFEA states you can move eggs/sperm/embryos into or out of the UK provided the import/export is to/from an HFEA-licensed clinic.
China: Expect higher uncertainty. China has national regulation around the management of human genetic resources (State Council release summary), and enforcement actions have targeted illegal ART practices including trade in gametes/zygotes/embryos and illegal surrogacy activity—factors that can complicate embryo export feasibility depending on the clinic, province, and documentation.
Surrogacy is widely described in academic analysis as prohibited by law in China, which is another reason China-origin embryo export cases require early China-qualified legal review.
Step six: U.S. customs + FDA admissibility review and delivery to the Manhattan clinic
FDA’s HCT/P import rule allows quarantine transport to the consignee while documentation is reviewed and FDA makes an admissibility decision.
FDA’s CBER import guidance highlights that HCT/P entry may require the establishment registration number (HRN) and an affirmation of compliance with 21 CFR Part 1271.
CBP guidance emphasizes importers’ responsibility for accurate documentation/labels/
Step seven: Manhattan intake, storage, and frozen embryo transfer (FET)
Once accepted, the Manhattan clinic performs intake/verification, transfers embryos into its storage system, and schedules the gestational carrier’s FET cycle.
A Manhattan pricing anchor: NYU Langone Fertility Center lists FET starting at $5,500, inclusive of ultrasound monitoring, embryo thaw/prep, and embryo transfer (medications excluded).
Eligibility criteria, common failure points, and timelines
Eligibility criteria (what makes a case workable in practice)
Identifiable donors and records: Because donor eligibility for embryos is required for both the oocyte and semen donor, cases are generally smoother when the genetic contributors are clearly documented, reachable for screening/testing, and the origin clinic can provide complete lab records.
Receiving clinic willing to accept the risk profile: FDA’s exceptions and labeling pathways exist, but the clinic still must decide whether it will accept the case under its compliance policy and medical director review.
Document quality + translation readiness: FDA requires records to be retained and translated to English with a translator authenticity statement, making documentation quality and translation logistics a real gating item.
Common failure points (what causes delays or outright refusal)
Timing mismatch for donor testing specimens: FDA’s donor testing regulation specifies timing windows around recovery (generally within 7 days; oocytes up to 30 days pre‑recovery). If the origin clinic did not perform testing within these windows, the Manhattan clinic may require exception labeling and still decline.
Inability to use CLIA + FDA donor-screening assays: FDA requires testing using FDA‑licensed/approved/cleared donor screening tests and performance by CLIA‑certified labs. If donor testing cannot be completed in this compliant manner (logistically or due to donor inaccessibility), remediation may fail.
Border friction from poor documentation: CBP states many biological materials imports arrive with missing/conflicting documentation; this increases the risk of holds and missed shipping windows.
China export feasibility issues: China’s regulatory environment and enforcement actions around illegal ART/surrogacy and embryo trade can make export slow or impossible depending on the case.
Timelines (what’s “regulatory time” vs “project time”)
Regulatory time windows (hard rules):
- Donor specimen timing: at recovery or within 7 days pre/post recovery; oocytes up to 30 days pre‑recovery.
- Anonymous semen donor retesting: at least 6 months after donation (does not apply to directed reproductive donors).
- Labeling when screening/testing wasn’t performed at recovery/cryopreservation but done later: FDA requires specific advisory labeling language.
Practical project timelines (estimates; vary by country and clinic policy):
- Europe → Manhattan, “typical”: 6–12 weeks is a common planning range for international shipments due to permits/authorizations and coordination complexity (estimate anchored to published international transport timelines by region).
- China → Manhattan, “typical”: 8–12+ weeks is common, with longer tails where export approvals are uncertain (estimate anchored to published Asia transport timelines, plus China-specific compliance uncertainty).
- Absolute fastest cases happen when the origin clinic’s records are complete, translation is immediate, donors are available for compliant testing within days, and the receiving clinic acceptance decision is rapid. (Clinic-specific; do not promise timelines.)
Pricing and itemized cost tables
How to read these tables
- Sourced figures are numbers explicitly published by referenced organizations (e.g., NYC clinic price list; courier cost guide).
- Estimated ranges are budgeting ranges compiled from market cost guides and typical line-items; exact quotes vary widely by route, urgency, and clinic policy. Surrogacy4All should publish these as “planning estimates” and always confirm with written quotes.
Europe → Manhattan (USD)
| Line item | What it covers | Range (USD) | Source status |
|---|---|---|---|
| Retrospective donor testing (CLIA lab + FDA donor screening assays) | Required infectious disease testing under FDA donor testing rules (HIV‑1/2, HBV, HCV, syphilis; plus CT/NG for reproductive donors), performed using FDA‑licensed/approved/cleared donor screening tests and by a CLIA-certified lab. | $800–$2,500 per genetic contributor (often 2 donors) | Estimate (regulatory requirements sourced; pricing varies by lab) |
| Donor eligibility documentation prep (“remediation packet”) | Coordinated donor screening record review + clinic-facing summary; donor eligibility determination must be documented by a responsible person. | $1,200–$3,500 | Estimate |
| Translation + translator authenticity statement | FDA requires records in English or translated to English with an authenticity statement; plus certified translation/notarization/ |
$300–$1,800 | Mixed: translation per doc ranges sourced from a courier cost guide; case totals vary |
| Courier service (Europe lane) | Hand-carry or managed freight, dry shipper, tracking/logging, chain of custody (depends on courier). Published “typical costs by region” for Europe are in the $3,000–$5,000 range for courier service alone. | $3,000–$6,500 | Mixed: region lane costs sourced; route-specific uplift estimated |
| Dry shipper rental + monitoring + insurance | Dry vapor shipper rental, temperature logging, declared value coverage. Cryoport describes extended hold time up to 10 days for its shipper. | $400–$1,800 | Mixed: hold-time sourced; premiums vary |
| Export permits / origin authorizations | EU/UK authorization processes vary by country; UK example requires HFEA-licensed clinic involvement. Published UK/EU authorization line items often budget $400–$800. | $400–$1,500 | Mixed |
| U.S. customs broker + entry filing + FDA admissibility handling | Preparing entry data, broker fees, and managing communications; FDA import guidance references HRN + affirmation of compliance; CBP warns documentation errors cause delays. | $300–$1,500 | Mixed: broker fee ranges sourced from cost guide; U.S. entry complexity varies |
| Manhattan clinic acceptance/intake/handling | Receiving clinic intake and compliance review; many clinics charge receiving and administrative fees (varies). Courier cost guide suggests receiving intake $300–$800 as common, but Manhattan programs can be higher. | $800–$3,500 | Estimate |
| Interim storage (Manhattan) | Storage while waiting for GC cycle timing; cost guide cites first-year storage $500–$1,200 as typical. | $500–$1,800 | Mixed |
| Frozen embryo transfer (FET) into GC (Manhattan) | Example published NYC anchor: FET starts at $5,500 (excludes meds). | $5,500–$8,500 | Sourced anchor + estimate |
| Estimated subtotal (Europe → Manhattan) | $13,000–$33,100 | Mixed | |
| Contingency: remediation fails / clinic declines | Additional compliance review, re‑shipping, alternate destination, or creating new embryos. A NYC anchor lists IVF without PGT at $17,600 and with PGT at $20,400 (meds excluded). | +$1,500–$6,000 or +$17,600–$20,400+ | Sourced anchor + estimate |
China → Manhattan (USD)
| Line item | What it covers | Range (USD) | Source status |
|---|---|---|---|
| Retrospective donor testing (CLIA lab + FDA donor screening assays) | Same FDA requirements: specified tests + CLIA + FDA‑labeled donor screening assays. | $800–$2,500 per genetic contributor | Estimate |
| Donor eligibility documentation prep (“remediation packet”) | Donor screening records + documented donor eligibility determination by responsible person. | $1,200–$3,800 | Estimate |
| Translation/notarization | Translation to English with authenticity statement; China-origin cases often require more documents and certified translation. | $600–$2,800 | Mixed |
| Courier service (Asia lane) | Courier cost guide shows Asia courier service often $4,000–$6,500; China routes can be higher depending on city and constraints. | $4,000–$8,000 | Mixed |
| Dry shipper + monitoring + insurance | Extended-hold LN2 shipper (e.g., up to 10 days for Cryoport Express); declared value coverage. | $500–$2,200 | Mixed |
| Export approvals/feasibility buffer (China) | China’s HGR regulation environment and enforcement actions around illegal ART/surrogacy and embryo trade can add time/cost uncertainty; plan for additional compliance/legal support. | $1,000–$6,000 | Estimate (risk driver sourced, dollars estimated) |
| U.S. customs broker + entry filing + FDA admissibility handling | Filing + admissibility review coordination; FDA import guidance references HRN + affirmation; CBP stresses documentation. | $400–$2,000 | Mixed |
| Manhattan clinic acceptance/intake/handling | Intake/compliance review and handling (clinic-specific). | $800–$3,500 | Estimate |
| Interim storage (Manhattan) | Storage while waiting for GC timing. | $500–$1,800 | Mixed |
| Frozen embryo transfer (FET) into GC (Manhattan) | NYC anchor: FET starts at $5,500 (excludes meds). | $5,500–$8,500 | Sourced anchor + estimate |
| Estimated subtotal (China → Manhattan) | $15,100–$40,800 | Mixed | |
| Contingency: export blocked / remediation fails | Could require alternative destination (e.g., Toronto) or creating new embryos (NYC IVF price anchors: $17,600–$20,400+ plus meds). | +$2,000–$10,000 or +$17,600–$20,400+ | Sourced anchor + estimate |
Canada mini option (Toronto; Health Canada framework) — pricing + notes
Regulatory/structural note (must be stated plainly): Canada’s Assisted Human Reproduction Act prohibits paying a surrogate and prohibits accepting consideration for arranging surrogacy services. This is a major difference from U.S. compensated surrogacy programs.
Canada also has a federal regulatory framework governing safety for donor sperm and ova (Safety of Sperm and Ova Regulations; current as of 2026-02-18 per consolidated regs listing), with detailed requirements around donor suitability and related processes.
| Canada (Toronto) line item | What it covers | Range (USD) | Source status |
|---|---|---|---|
| Toronto clinic FET fee | TRIO Fertility publishes Frozen Embryo Transfer at $2,850 (CAD), with meds typically $1,000–$3,000+ (CAD). Convert to USD at prevailing FX at time of quote. | ~Varies with FX | Sourced (CAD), USD varies |
| Medications for FET | TRIO Mississauga notes medication can range $1,000–$3,000+ (CAD) depending on protocol. | ~Varies with FX | Sourced (CAD), USD varies |
| Clinic handling fee for transfers (example) | One Fertility publishes a handling fee of $550 + HST (CAD) for sperm/oocyte/embryo transfer to another clinic, plus outgoing courier costs. | ~Varies with FX | Sourced (CAD), USD varies |
| Shipping to Toronto | Similar cost components (courier + shipper + permits + translations). Use Europe/Asia lane shipping ranges as planning estimates. | $4,000–$10,000 | Estimate |
Recommended workflows and a decision tree
Recommended workflows (what Surrogacy4All should operationalize)
Workflow A: “Manhattan-first feasibility” (recommended default)
Start with a Manhattan receiving clinic pre‑review and a written list of required documents/testing. This aligns your entire plan with the reality that FDA requires donor eligibility for embryo transfer (with limited exceptions) and that import entry may require HCT/P establishment registration number and compliance affirmations.
Workflow B: “Dual-track Manhattan + Toronto” (recommended for China cases or weak documentation)
Because China-origin cases can face export feasibility issues and enforcement headwinds around illegal ART/surrogacy and embryo trade, consider concurrently evaluating whether Toronto can accept the embryos under Canadian frameworks as a contingency.
Workflow C: “Create new embryos in North America” (when remediation is structurally impossible)
If donor identity/testing is impossible or the receiving clinic refuses exception/labeling pathways, plan for embryo re‑creation in a North American clinic with transparent pricing anchors.
Mermaid decision tree
Frequently asked questions
Q. Does the FDA “certify” IVF clinics?
A. FDA generally regulates reproductive tissues under HCT/P rules; establishments performing steps in HCT/P manufacturing must register and list their HCT/Ps with FDA (HCT/P establishment registration). Verification can be done via FDA’s HCTERS public query.
Q. What exactly makes a U.S. clinic refuse embryos from an overseas clinic?
A. Common reasons are missing donor eligibility documentation (screening/testing), inability to verify compliant testing methods/lab status (CLIA + FDA-labeled tests), poor chain-of-custody documentation, and uncertainty around exception/labeling pathways.
Q. What is “FDA remediation” in practice?
A. “FDA remediation” is not a single FDA-defined process; it is a practical coordination term for assembling the records, donor screening summary, compliant donor testing, translations, labeling plan, and clinic-facing documentation needed for a receiving clinic to accept an embryo transfer case. FDA’s donor eligibility requirements and labeling rules are the legal framework that dictates what must be addressed.
Q. Can you just ship the embryos first and fix paperwork later?
A. HCT/P import rules contemplate quarantine transport to the consignee while FDA reviews documentation and makes an admissibility decision; however, CBP underscores that missing/conflicting documentation is common and causes delays. In practice, most teams aim to finalize documentation before the shipment moves.
Q. Is Toronto a “backup plan” for U.S. surrogacy?
A. Toronto can be a clinical alternative for embryo transfer logistics, but Canada’s legal structure differs: Canada prohibits paying a surrogate and prohibits accepting consideration for arranging surrogacy. Clients must understand that this may change the overall journey design.